Cell Technology
-
-
Stem cell technology

-
The Symposium on Scientific and Technological Innovation Base of Private Enterprises in Shanghai and the Awarding Ceremony was successfully held on January 2, 2020. As a representative of sci-tech innovative private enterprises, Fosun Pharma was successfully listed as a candidate and awarded the title of Key Laboratory of Stem Cell Therapy Drugs in Shanghai! This is the first time that the Key Laboratory in Shanghai has been awarded to a private enterprise. Wu Qing, a member of the Standing Committee of Shanghai Municipal Committee and Vice Mayor, attended the symposium and awarded the license to scientific and technological innovation base to private enterprise.
Fosun Pharma, in collaboration with ReNeuron (UK), has jointly developed allogeneic stem cell therapy products for the treatment of blindness caused by post-stroke disability and retinitis pigmentosa in stroke patients and patients with rare retinal diseases causing blindness in huge need of treatment. There is no effective treatment for both indications worldwide. In order to promote industrialization, Fosun Pharma actively established its "Key Laboratory of Stem Cell Therapy Drugs" to fully promote global clinical studies, initiate technology transfer and enable stem cell therapy to benefit patients in China and around the world as soon as possible. Fosun Pharma will take these two stem cell technologies as the starting point and strive to build a sound stem cell technology platform in recent years. By introducing foreign advanced technology and talents, Fosun Pharma will integrate a team with stem cell technology as the core backbone and a comprehensive reserve of outstanding talents in technical links for the laboratory, create a unicorn company in stem cell industry and strive to reach the international leading level!
-
-
-
-
Cell therapy platform

-
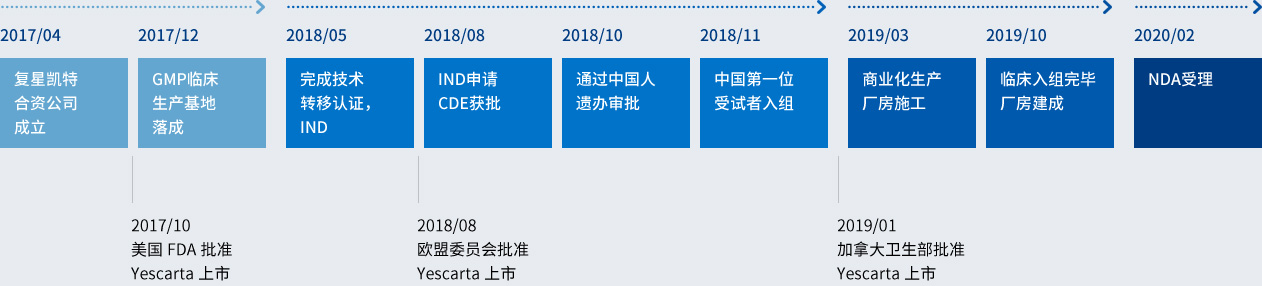
As a joint venture of Shanghai Fosun Pharmaceutical (Group) Co., Ltd and U.S. Kite Pharma, Fosun Kite Biotechnology Co., Ltd. (hereinafter referred to as "Fosun Kite") was established in April 2017 in Shanghai China. Taking a combined approach of internal R&D and external partnering/technology transfer, Fosun Kite is dedicated to the advancement of innovative cell therapy and its industrialization in China to benefit patients.
As a leading medical and health industry group in China, Fosun Pharma has abundant experience in pharmaceutical manufacturing and R&D, and has a number of products under research in the field of cancer treatment. Kite Pharma is a global leader in the research and development of T cell immunotherapy. The establishment of the joint venture will take advantage of the resource advantages of both sides to build a leading T cell immunotherapy industry platform in China and jointly provide safe and effective T cell immunotherapy products for Chinese cancer patients.
-

R&D
Core independent R&D platform
Strategic collaborative development partners
Development capabilities for commercial cell therapy products -

Production
An integrated supply chain for cell therapy products;
Rapid technology transfer capability of cell therapy products;
Localized production capacity in compliance with GMP, strict quality control of cell therapy products -
Clinical
Rapid generation of preclinical data;
Rich experience in clinical trials of cell therapy products;
Experience in registration of cell therapy products and translational medicine capabilities. -

Commercialization
Business model in line with the Chinese market
Academic promotion ability
Sales network
-
-
Gene Technology
Introduction to COVID-19 mRNA Vaccine Project
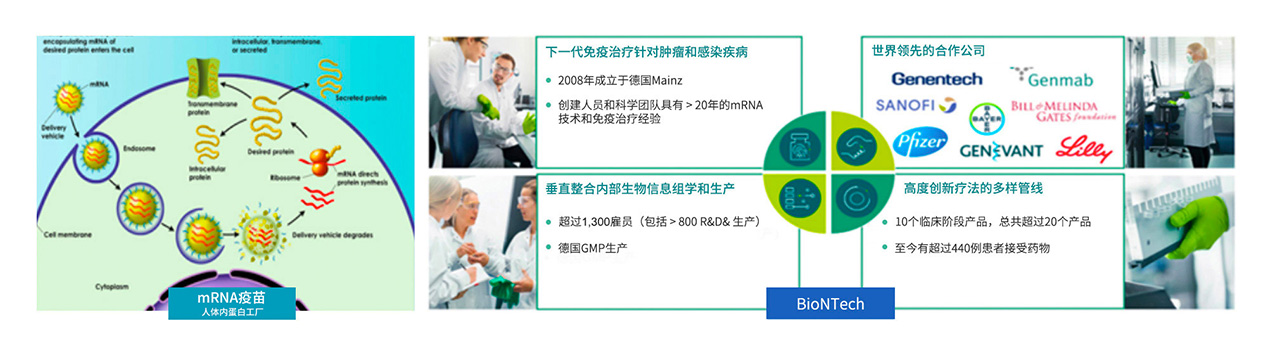
In early 2020, COVID-19 outbreaks spread around the world. Fosun Pharma actively investigated the progress of global vaccine research and development, sought international cooperation, and finally selected BioNTech, a leading German company in the field of mRNA vaccines, as a partner. On March 16, 2020, Fosun Pharma announced that it had signed a cooperation agreement with BioNTech for the authorization of clinical development and commercialization in mainland China and Hong Kong, Macao and Taiwan of the COVID-19 vaccines (including BNT162 vaccine candidate, etc.) developed by BioNTech through RNA technology platform.
Fosun and BioNTech jointly developed the mRNA vaccine against COVID-19 mRNA vaccine project
-
- Successful Collaboration 11 COVID-19 mRNA Vaccine Project
- Fosun's outlook on the epidemic and strong sense of social responsibility
- Advantages of mRNA vaccine
- BioNTech's leading technologies, R&D capacity and rapid response of Fosun Pharma
-
- mRNA Vaccine 11 COVID-19 mRNA Vaccine Project
- Short R&D cycle
- Dual mechanism of action: humoral/cellular immunity
- Potent, safe without adjuvant
- Simple process and rapid batch production
-
- BioNTech's leading technologies 111 COVID-19 mRNA Vaccine Project
- Comprehensive R&D capabilities of diverse mRNA technologies
- Highly efficient and safe LNP delivery
- Experience in mRNA trials in > 250 human subjects
- Preliminary clinical tumor vaccine efficacy High yield
- Broad immunotherapy platform
There are three different forms of the mRNA vaccine (BioNTech) introduced by Fosun, uridine mRNA, nucleoside-modified mRNA, and self-amplified mRNA. These three vaccine forms have varied advantages, as shown in the table below.
-
-
Uridine mRNA 11 contains three different forms (uRNA/BNT162 a1) 111 contains three different forms
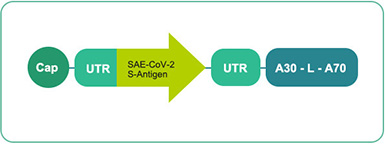
Clinical phase technology
Manufacturing and CMC processes in place -
Basis:
Endogenous adjuvant effect
Expected good tolerability and safety
Antibodies & strong T-cell response
-
-
-
Nucleoside-modified mRNA (modRNA/BNT162 b1&b2)
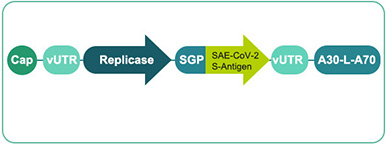
Clinical phase technology
Manufacturing and CMC processes in place -
Basis:
Less adjuvant effect
Expected good tolerability and safety
Strong antibody response & CD4 T cell response
-
-
-
Self-amplified mRNA (modRNA/BNT162 b1&b2)
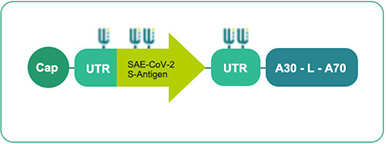
Clinical phase technology
GLOP toxicological data available
Manufacturing and CMC processes in place -
Basis:
Long-term protein expression
Expected good tolerability and safety
Highly possible good immune response after a single dose
Possible efficacy at very low vaccine dose
-
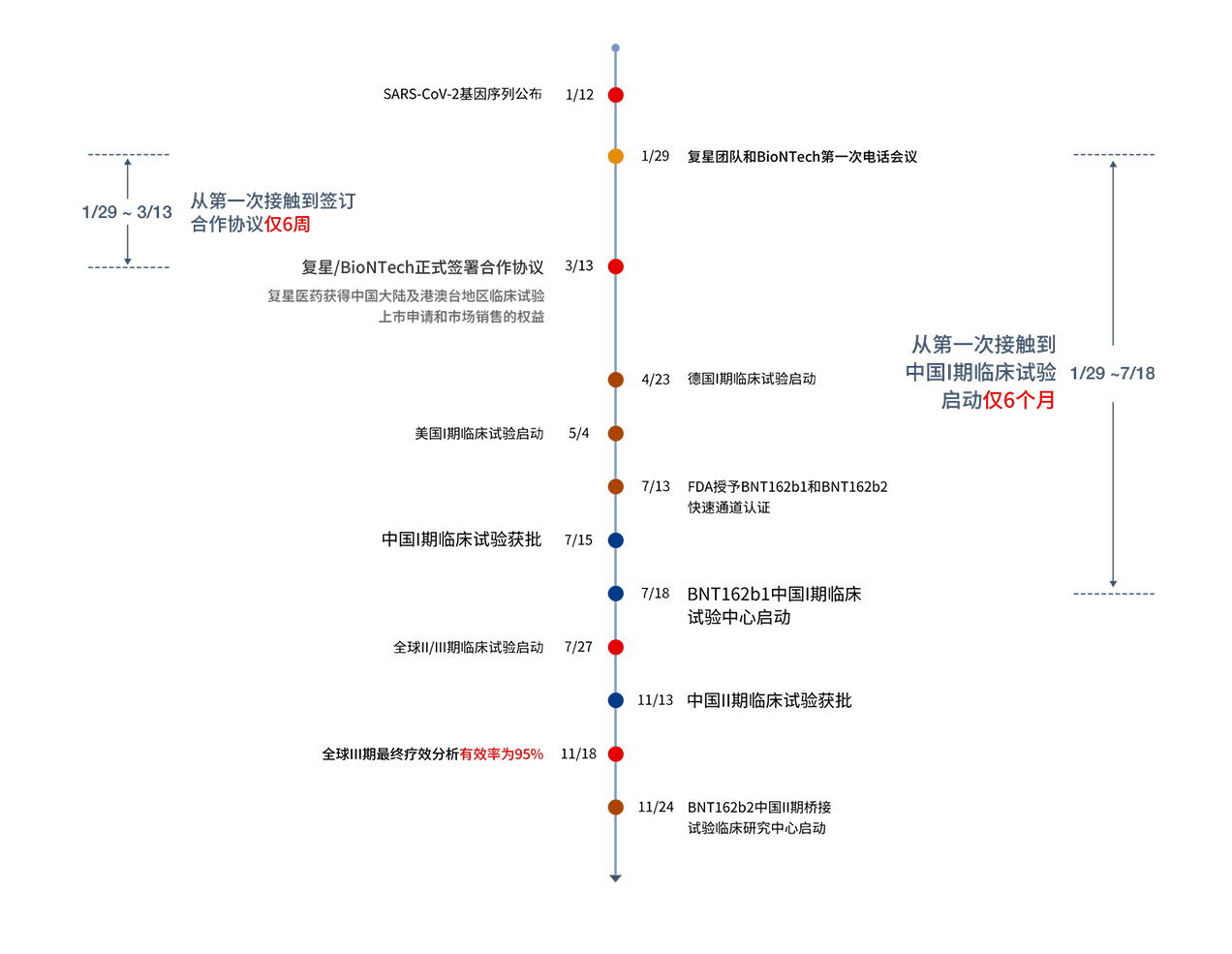
o fight against the epidemic, Fosun Pharma and BioNTech work together worldwide for mRNA vaccine research and development:
In April 2020, Fosun Pharma and the Institute of Laboratory Animals Science of CAMS & PUMC initiated the mouse challenge test and timely shared the test results with partners.
In April and May 2020, BioNTech initiated a phase I/IIa clinical trial of BNT162b1/b2.
On July 18, 2020, Fosun Pharma initiated a phase I clinical trial of BNT162b1 in China on the basis of partner data sharing.
On July 27, 2020, BNT162b2 was finally selected for a global phase IIb/III clinical trial after multi-party evaluations.
On November 9, the results of the first interim analysis showed that the efficacy of BNT162b2 was over 90% 7 days after completion of the vaccination procedure.
On November 13, Fosun Pharma was approved to carry out a phase II bridging trial of BNT162b2 in China.
On November 18, the final efficacy analysis of the global phase III clinical trial showed that BNT162b2 met all the primary efficacy endpoints of the study, with an effective rate of 95%.
On November 24, Fosun Pharma held a phase II clinical trial initiation meeting in Taizhou, Jiangsu Province
Fosun Pharma is making every effort to complete the clinical development, registration and marketing of the vaccine as soon as possible.
Timeline for Collaborative Development of mRNA Vaccine by BioNTech/Fosun
Introduction to a Cancer Vaccine, SurVaxM
SurVaxM is a peptide mimic immunotherapeutic vaccine (immunotherapy) that targets survivin, a cell-survival protein present inside or on the surface of 90% of glioblastoma and many other cancer cells. SurvAxM has a dual mechanism of action of stimulating T cell immunity, as well as antibody-mediated inhibition of the survivin pathway, which can control tumor growth and prevent or delay tumor recurrence.
This project was introduced with an exclusive license from MimiVax, LLC (USA) in November 2019 for the clinical development, registration and marketing in mainland China, Hong Kong and Macao for the indications of glioblastoma and other tumors.
-
-
VONSUN

-
Oncolytic virotherapy (OV) is a novel tumor therapy that kills tumors by selectively infecting tumor cells with viruses. Oncolytic virus is a kind of tumor-killing virus with replication ability, which can selectively infect tumor cells and replicate in tumor cells and then kill tumor cells, and stimulate the body to produce a specific anti-tumor immune response. Only a few viruses in nature can naturally cause oncolysis, and most of them require genetic modification to achieve the effects of targeted infection of tumor cells and treatment. As an important part of tumor immunity, it has the following advantages over other tumor immunotherapies:
1) Acting on multiple links of tumor immunity, broad-spectrum anti-cancer effect, effective for recurrent and metastatic tumors;
2) Reliable safety, fewer side effects, and no dose-limiting toxicities observed. At present, a number of drugs worldwide are under phase III clinical trials, and have shown good or even revolutionary therapeutic effects in clinical trials, especially in some cancers with very low survival such as glioma, demonstrating great potential, and even achieving lasting disease remission in some patients, which brings forward a promising way for cancer treatment. At present, Fosun Pharma has initiated the development of oncolytic virus products, which is in the preclinical research stage.
-