全球研发|复星医药子公司复宏汉霖7款FIC/BIC创新管线即将亮相 AACR 2026

全球肿瘤学领域最具影响力的学术盛会之一——美国癌症研究协会年会(AACR 2026)将于2026年4月17日至22日在美国圣地亚哥举行。本届AACR大会上,复星医药子公司复宏汉霖将围绕HLX43(PD-L1 ADC)、HLX316(B7-H3×唾液酸酶融合蛋白)、HLX3902(STEAP1×CD3×CD28三特异性TCE)及依托Hanjugator™等自主平台开发的多款新型ADC(EGFRxcMET双抗ADC HLX48、HER2双表位ADC HLX49、ADAM9 ADC HLX402、ALPP/ALPPL2 ADC HLX85),集中展示7款具有“同类首创”或“同类最优”潜力的早期创新资产,覆盖ADC、TCE和新型糖免疫调节融合蛋白在内的前沿赛道,全面呈现其差异化、多平台驱动的创新研发矩阵。
七款FIC/BIC创新资产集中亮相
-
HLX43(PD-L1 ADC)
广谱抗肿瘤活性的创新作用机制
摘要标题:临床前研究揭示蛋白酶介导的载荷释放及肿瘤免疫/免疫调节效应在HLX43的抗肿瘤活性中发挥重要作用
Preclinical studies reveal that protease-mediated payload release and immuno-oncology/immunomodulatory effects contribute to the antitumor activity of HLX43
分会场标题:Mechanisms of Anticancer Drug Action
展示形式:壁报
摘要编号:5704
展示时间:美国东部时间4月21日 2:00 PM-5:00 PM
展示地点:第12区,展板#20
-
HLX316(B7H3×唾液酸酶融合蛋白)
潜在FIC的肿瘤免疫调控与糖免疫检查点分子
摘要标题:一款靶向B7-H3的人唾液酸酶融合蛋白,可协同增强先天及适应性抗肿瘤免疫反应
Human sialidase-armed anti-B7-H3 antibody that enhances innate and adaptive antitumor immune responses分会场标题:Overcoming Microenvironmental and Delivery Barriers in Cancer Therapy
展示形式:壁报
摘要编号:7158
展示时间:美国东部时间4月22日 9:00 AM-12:00 PM
展示地点:第15区,展板#15
-
HLX3902(STEAP1×CD3×CD28三特异性TCE)
前列腺癌STEAP1xCD3xCD28 TCE
摘要标题:一款新型CD3×CD28×STEAP1三特异性T细胞衔接器在前列腺癌中展现强效、持久的抗肿瘤应答
A novel anti-CD3×CD28×STEAP1 tri-specific T-cell engager with enhanced and durable antitumor responses in prostate cancer
分会场标题:Redefining Targeted Therapy: Bispecific T-Cell Engagers and Antibody-Drug Conjugates 2
展示形式:壁报
摘要编号:5397
展示时间:美国东部时间4月21日 9:00 AM-12:00 PM
展示地点:第48区,展板#10
-
Hanjugator™自主ADC平台及HLX48(EGFR×cMET ADC)
更安全有效的差异化双抗ADC
摘要标题:Hanjugator喜树碱平台:高效低毒设计最大化抗体潜能,孵化潜在同类最优的EGFR/cMET双抗ADC
Hanjugator camptothecin platform: Effective, low-toxicity design maximizing antibody functionality and delivering potential best-in-class EGFR/cMet bispecific antibody-drug conjugate
分会场标题:Tumor Microenvironment, Multispecifics, and Immunomodulation
展示形式:壁报
摘要编号:5856
展示时间:美国东部时间4月21日 2:00 PM-5:00 PM
展示地点:第17区,展板#25
-
HLX49(HER2双表位ADC)
潜在BIC的新型HER2双表位ADC
摘要标题:一款潜在同类最优的HER2双表位ADC,通过高效低毒设计最大化抗体潜能
A best-in-class HER2xHER2 novel biparatopic antibody-drug conjugate with an efficacious, low-toxicity design that maximizes antibody functionality
分会场标题:Antibody Technologies and Platforms 2
展示形式:壁报
摘要编号:4395
展示时间:美国东部时间4月21日 9:00 AM-12:00 PM
展示地点:第11区,展板#3
-
HLX402(ADAM9 ADC)
潜在BIC的新兴肿瘤靶点ADC
摘要标题:靶向ADAM9(ADAM金属肽酶结构域9)的喜树碱ADC临床前数据亮眼,具备同类最优潜力
Preclinical characterization of a potential best-in-class camptothecin-based antibody-drug conjugate targeting ADAM metallopeptidase domain 9
分会场标题:Antibody Technologies and Platforms 2
展示形式:壁报
摘要编号:4418
展示时间:美国东部时间4月21日 9:00 AM-12:00 PM
展示地点:第11区,展板#26
-
HLX85(ALPP/ALPPL2 ADC)
潜在FIC的新兴肿瘤靶点ADC
摘要标题:潜在同类首个靶向ALPP/ALPPL2的喜树碱ADC展现强效抗肿瘤活性和卓越耐受性
Development of a first-in-class camptothecin-based antibody-drug conjugate targeting ALPP/ALPPL2 with potent antitumor activity and excellent tolerability
分会场标题:Antibody Technologies and Platforms 2
展示形式:壁报
摘要编号:4419
展示时间:美国东部时间4月21日 9:00 AM-12:00 PM
展示地点:第11区,展板#27
多平台协同发力,驱动早期创新管线加速孵化
复宏汉霖此次带来的多项创新研究成果,将集中展示公司在抗体药物偶联物(ADC)、免疫细胞衔接器(如TCE)以及肿瘤免疫治疗(IO)等领域的领先研发能力。
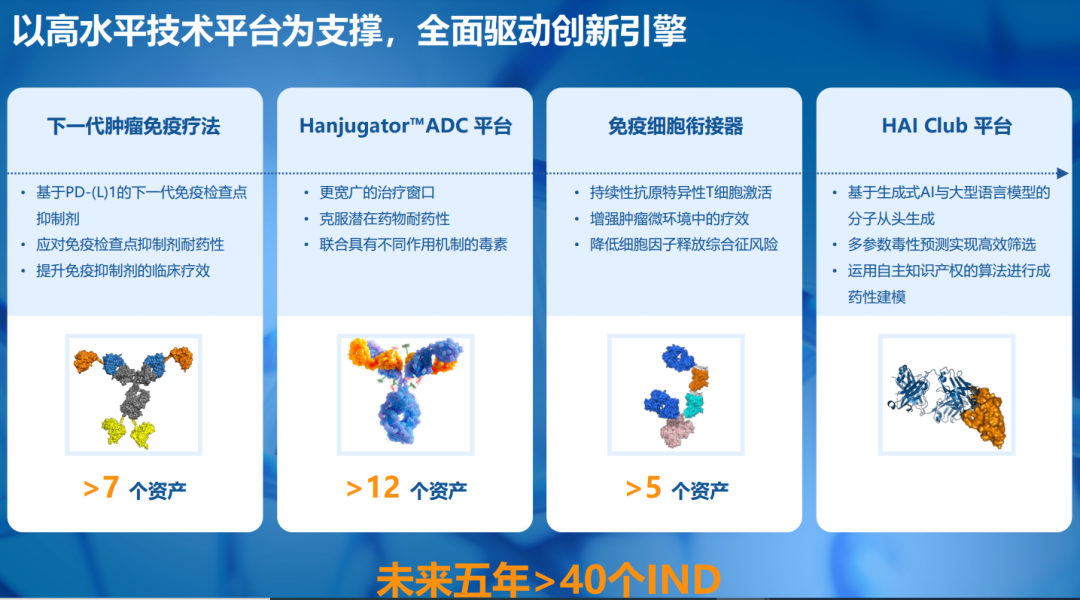
目前,从早期靶点筛选、验证,候选分子设计与优化,到系统性地临床前开发,公司已搭建起包括PD-(L)1为核心的免疫检查点抑制剂平台、免疫细胞衔接器平台(如多特异性TCE平台)、Hanjugator™ ADC 平台、AI驱动的一站式早期研发平台HAI Club在内的多维创新平台矩阵,不仅保障了单个项目的研发质量与效率,更关键的是为打造具备全球竞争力的中长期创新管线提供了可持续的系统能力支撑,从而能够持续、高效地将前沿科学发现转化为具有临床价值的候选药物。未来五年内,公司预计将有超过40项新的临床研究申请获批。
内容来源:AACR官网
关于复宏汉霖
复宏汉霖(2696.HK)是一家国际化创新生物制药企业,致力于为全球患者提供高品质、可负担的生物药,产品覆盖肿瘤、自身免疫疾病、眼科疾病等领域。自2010年成立以来,公司已构建涵盖全球研发、临床、注册、生产及商业化的全产业链平台,拥有全球员工近4,000人,并在中国、美国和日本等多地设有运营及分支机构。依托生物类似药形成的稳健现金流反哺创新研发,复宏汉霖正稳步迈入“全球化2.0”阶段,持续打造可复制、可持续的全球增长模式。截至2026年初,公司共有10款产品在全球60个国家和地区获批上市,其中7款已在中国获批。在欧美主流生物药市场,复宏汉霖亦取得多项里程碑式突破,已有4款产品获得美国FDA批准、4款产品获得欧盟EMA上市授权,充分体现了公司在研发体系、质量管理及生产能力方面已全面对标国际最高标准。
在创新驱动方面,复宏汉霖依托上海、美国等多地协同布局的研发体系,构建了多元化、平台化的创新技术矩阵,覆盖免疫检查点抑制剂、免疫细胞衔接器(包括多特异性TCE)、抗体偶联药物(ADC)以及AI驱动的早期研发平台等前沿方向。目前,公司拥有50余项处于早期阶段的创新资产,其中约70%具备同类最佳(Best-in-Class)潜力,并在全球同步推进30余项临床研究。核心产品H药 汉斯状®(斯鲁利单抗,欧洲商品名:Hetronifly®)作为全球首个获批一线治疗小细胞肺癌的抗PD-1单抗,正加速全球布局,已在全球40余个市场获批上市;同时,多款潜力创新资产,包括PD-L1 ADC HLX43及新表位HER2单抗HLX22正全面推进全球关键性临床研究。依托通过中、欧、美三地GMP认证的生产体系,复宏汉霖已建成总产能达84,000升的生物药生产平台,形成覆盖全球六大洲的稳定供应网络。未来,复宏汉霖将始终坚持以患者为中心,聚焦未满足的临床需求,持续推动创新成果向临床价值与患者可及转化,在全球生物医药创新生态中创造长期而稳健的价值。
Henlius to Showcase Seven FIC/BIC Innovative Assets at AACR 2026, Highlighting Accelerated Early-Stage Innovation
The American Association for Cancer Research (AACR) Annual Meeting 2026, one of the most influential academic events in global oncology, will be held from April 17 to 22, 2026, in San Diego, USA. At this year’s AACR conference, Henlius will showcase seven early-stage innovative assets with first-in-class (FIC) or best-in-class (BIC) potential. These include HLX43 (PD-L1 ADC), HLX316 (B7-H3×sialidase fusion protein), HLX3902 (STEAP1×CD3×CD28 trispecific T-cell engager), as well as multiple novel ADC candidates developed based on proprietary platform Hanjugator™, including the EGFR×cMET bispecific ADC HLX48, HER2 biparatopic ADC HLX49, ADAM9 ADC HLX402, and ALPP/ALPPL2 ADC HLX85. Together, these innovative assets span cutting-edge modalities including ADCs, TCE, and novel glyco-immunomodulatory fusion protein, highlighting Henlius’ differentiated, multi-platform-driven innovation matrix.
________________________________________
Seven Innovative Assets with FIC/BIC Potential
HLX43 (PD-L1 ADC)
•Title: Preclinical studies reveal that protease-mediated payload release and immuno-oncology/immunomodulatory effects contribute to the antitumor activity of HLX43
•Session Title: Mechanisms of Anticancer Drug Action
•Format: Poster Session
•Abstract Number: 5704
•Date and Time: April 21, 2026, 2:00 PM-5:00 PM ET
•Location: Poster Section 12, Board #20
________________________________________
HLX316 (B7-H3 × Sialidase Fusion Protein)
•Title: Human sialidase-armed anti-B7-H3 antibody that enhances innate and adaptive antitumor immune responses
•Session Title: Overcoming Microenvironmental and Delivery Barriers in Cancer Therapy
•Format: Poster Session
•Abstract Number: 7158
•Date and Time: April 22, 2026, 9:00 AM-12:00 PM ET
•Location: Poster Section 15, Board #15
________________________________________
HLX3902 (STEAP1 × CD3 × CD28 Tri-specific TCE)
•Title: A novel anti-CD3×CD28×STEAP1 tri-specific T-cell engager with enhanced and durable antitumor responses in prostate cancer
•Session Title: Redefining Targeted Therapy: Bispecific T-Cell Engagers and Antibody-Drug Conjugates 2
•Format: Poster Session
•Abstract Number: 5397
•Date and Time: April 21, 2026, 9:00 AM-12:00 PM ET
•Location: Poster Section 48, Board #10
________________________________________
Hanjugator™ Platform & HLX48(EGFR×cMET ADC)
•Title: Hanjugator camptothecin platform: effective, low-toxicity design maximizing antibody functionality and enabling a potential best-in-class EGFR/cMet bispecific ADC
•Session Title: Tumor Microenvironment, Multispecifics, and Immunomodulation
•Format: Poster Session
•Abstract Number: 5856
•Date and Time: April 21, 2026, 2:00 PM-5:00 PM ET
•Location: Poster Section 17, Board #25
________________________________________
HLX49 (HER2xHER2 Biparatopic ADC)
•Title: A best-in-class HER2xHER2 biparatopic antibody-drug conjugate with an efficacious, low-toxicity design that maximizes antibody functionality
•Session Title: Antibody Technologies and Platforms 2
•Format: Poster Session
•Abstract Number: 4395
•Date and Time: April 21, 2026, 9:00 AM-12:00 PM ET
•Location: Poster Section 11, Board #3
________________________________________
HLX402 (ADAM9 ADC)
•Title: Preclinical characterization of a potential best-in-class camptothecin-based antibody-drug conjugate targeting ADAM metallopeptidase domain 9
•Session Title: Antibody Technologies and Platforms 2
•Format: Poster Session
•Abstract Number: 4418
•Date and Time: April 21, 2026, 9:00 AM-12:00 PM ET
•Location: Poster Section 11, Board #26
________________________________________
HLX85 (ALPP/ALPPL2 ADC)
•Title: Development of a first-in-class camptothecin-based antibody-drug conjugate targeting ALPP/ALPPL2 with potent antitumor activity and excellent tolerability
•Session Title: Antibody Technologies and Platforms 2
•Format: Poster Session
•Abstract Number: 4419
•Date and Time: April 21, 2026, 9:00 AM-12:00 PM ET
•Location: Poster Section 11, Board #27
________________________________________
Integrated Platforms Driving Sustainable Innovation
The findings to be presented at AACR 2026 underscore Henlius’ expanding innovation footprint across ADCs, TCEs, and immuno-oncology(IO).
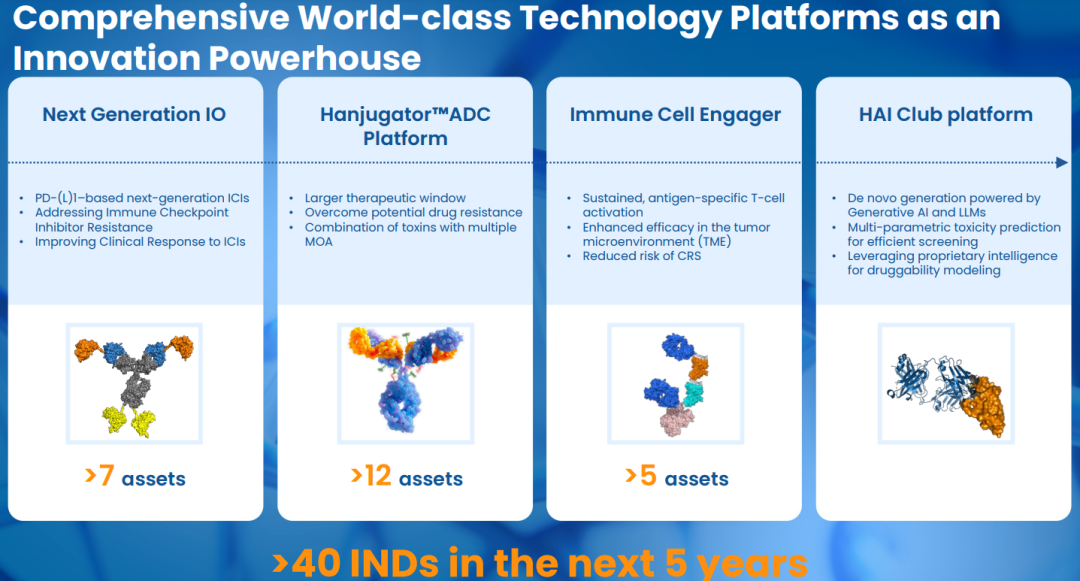
Henlius has established an integrated R&D system spanning target identification and validation, candidate design and optimization, and systematic preclinical development. This system is supported by a multi-dimensional technology platform framework, including a PD-(L)1-centred immune checkpoint inhibitor platform, immune cell engager platforms such as multi-specific T-cell engagers (TCEs), the Hanjugator™ ADC platform, and the AI-driven one-stop early discovery platform HAI Club. Together, these platforms support the efficient advancement of individual programs and contribute to the development of a differentiated and globally competitive pipeline. By enabling the translation of scientific discoveries into clinical-stage candidates, Henlius continues to advance its early-stage innovation capabilities. Looking ahead, the company expects to submit more than 40 investigational new drug (IND) applications over the next five years.
About Henlius
Shanghai Henlius Biotech, Inc. (2696.HK) is a global, innovation-driven biopharmaceutical company committed to delivering high-quality, affordable biologic therapies to patients worldwide. The Company focuses on major disease areas including oncology, autoimmune diseases, and ophthalmic diseases. Founded in 2010, Henlius has established an integrated, end-to-end biopharmaceutical platform encompassing global R&D, clinical operations, regulatory affairs, manufacturing, and commercialisation. The Company employs nearly 4,000 people globally and operates across multiple regions, including China, the United States, and Japan. Leveraging the stable cash flow generated from its biosimilar portfolio to support innovation, Henlius is steadily advancing into its “Globalisation 2.0” phase, building a scalable and sustainable global growth model. As of early 2026, Henlius has achieved regulatory approvals for 10 products across 60 countries and regions worldwide, including seven approvals in China. The Company has also reached multiple milestones in major biopharmaceutical markets, with four products approved by the U.S. Food and Drug Administration (FDA) and four products authorized by the European Medicines Agency (EMA), reflecting its globally aligned R&D capabilities, quality systems, and manufacturing standards.
Driven by innovation, Henlius has built a diversified, platform-based technology ecosystem through coordinated R&D efforts across Shanghai, the United States, and other regions. Its innovation platforms span immune checkpoint inhibitors, immune cell engager technologies (including multispecific T cell engagers), antibody-drug conjugates (ADCs), and AI-enabled early discovery platforms. The Company currently has more than 50 early-stage innovative assets, approximately 70% of which are expected to be best-in-class, with over 30 clinical trials ongoing globally. Henlius’ core product, serplulimab (trade name: Hetronifly® in Europe), is the world’s first anti–PD-1 mAb approved for first-line treatment of small cell lung cancer and has been approved in more than 40 markets worldwide with an accelerated globalisation process. In parallel, multiple high-potential innovative assets—including the PD-L1 ADC HLX43 and the novel epitope anti-HER2 mAb HLX22—are advancing through global pivotal clinical development. Supported by a biologics manufacturing network with a total capacity of 84,000L and GMP certifications from regulatory authorities in China, Europe, and the United States, Henlius has established a stable global supply system serving six continents. Guided by a patient-centred mission, Henlius remains focused on addressing unmet medical needs and translating scientific innovation into meaningful clinical value and patient access, contributing sustainably to the global biopharmaceutical ecosystem.
To learn more about Henlius, visit https://www.henlius.com/en/ and connect with us on LinkedIn at https://www.linkedin.com/company/henlius/.
联系方式

媒体:pr@fosunpharma.com
投资人:ir@fosunpharma.com










