全球研发|复星医药子公司复宏汉霖CDH17 ADC临床前数据首次发布:彰显消化道肿瘤领域差异化优势

近日,复星医药子公司复宏汉霖(2696.HK)于ADC Asia Congress 2026上首次披露其自主研发的靶向CDH17的抗体药物偶联物(ADC)HLX403的临床前研究数据。这款基于复宏汉霖自主知识产权Hanjugator™ ADC平台开发的候选分子展现出多维度的差异化优势,有望为消化道肿瘤的治疗提供一款更优的ADC候选分子。


左右滑动查看更多
消化道肿瘤(gastrointestinal cancers,GI cancers)是全球范围内疾病负担最重的癌症之一,每年新发病例超过500万,死亡病例约370万1,其中结直肠癌与胃癌长期位居发病率和死亡率前列,胰腺癌则以侵袭性强、预后差为特征,被视为致死率最高的实体瘤之一。尽管靶向治疗和免疫治疗的快速发展改善了部分患者的治疗结局,但相当比例的消化道肿瘤患者尤其是晚期或转移性患者,仍面临治疗选择有限、疗效持续性不足以及获得性耐药等挑战,提示该疾病领域仍存在显著的未满足临床需求2。近年来,抗体药物偶联物(ADC)凭借其精准的肿瘤杀伤机制,已成为消化道肿瘤领域的前沿研究方向及临床开发热点,靶向HER2、Claudin18.2、CDH17、c-MET等ADC药物研发进展迅速,显示出良好的抗肿瘤活性及可管理的安全性特征,有望进一步拓展消化道肿瘤患者的精准治疗策略3。

其中,Cadherin-17(CDH17)属于钙黏蛋白家族,通常在胃肠上皮细胞的紧密连接处表达。在胃癌、结直肠癌和胰腺癌等多类消化道肿瘤中,CDH17呈现高表达,且表达水平与患者的不良预后密切相关。由于CDH17在正常组织中表达受限,而在恶性肿瘤细胞表面暴露,这一选择性表达特征使其成为开发更精准ADC疗法的重要靶点4-5。
此次公布的多项临床前研究结果显示该分子具备:
1. 卓越的抗体性能与特异性
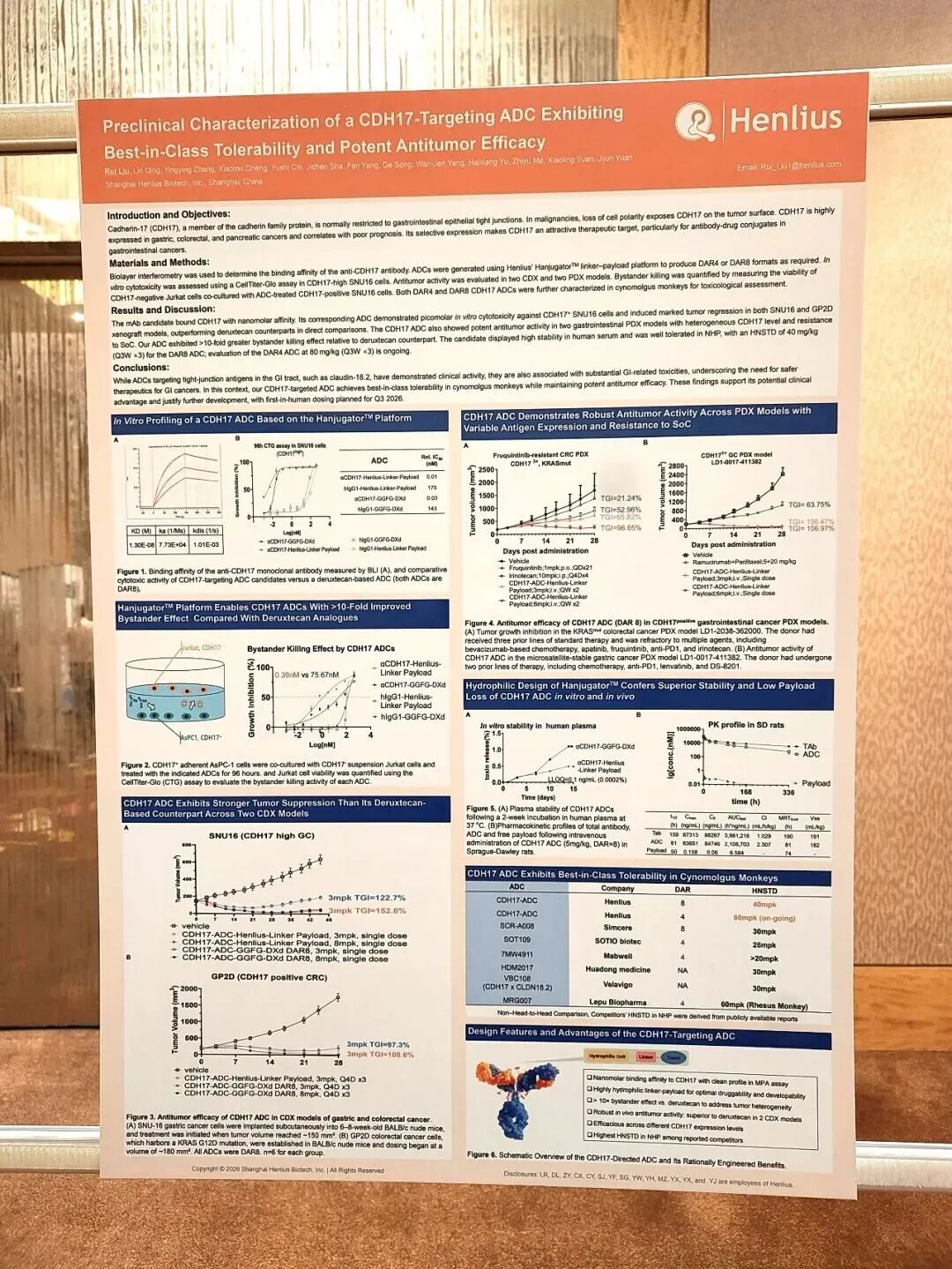
• 高亲和力与特异性: 该ADC使用的抗体对CDH17具有纳摩尔级(nanomolar)的结合亲和力;不会与钙粘蛋白家族的其他成员发生脱靶结合,且在MPA检测中表现干净。
2. 立足差异化的连接子-载荷(Linker-Payload)平台(HanjugatorTM)
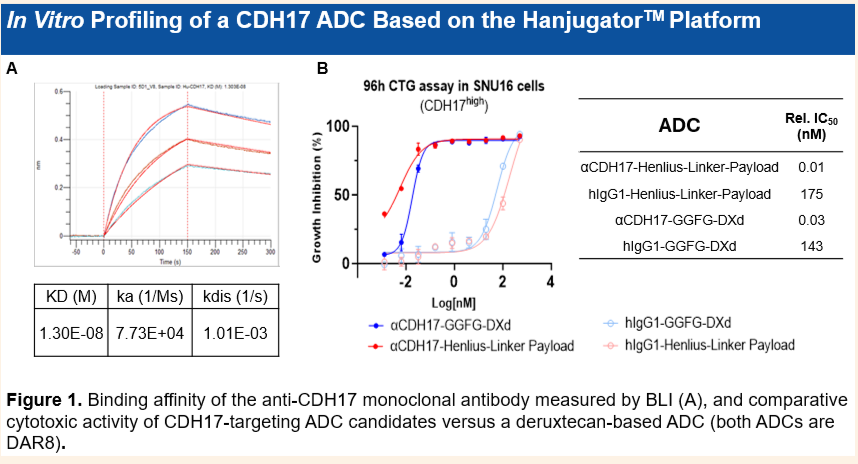
• 高亲水性设计: 采用自主研发的亲水性Linker-Payload平台,具有极佳的成药性和可开发性。
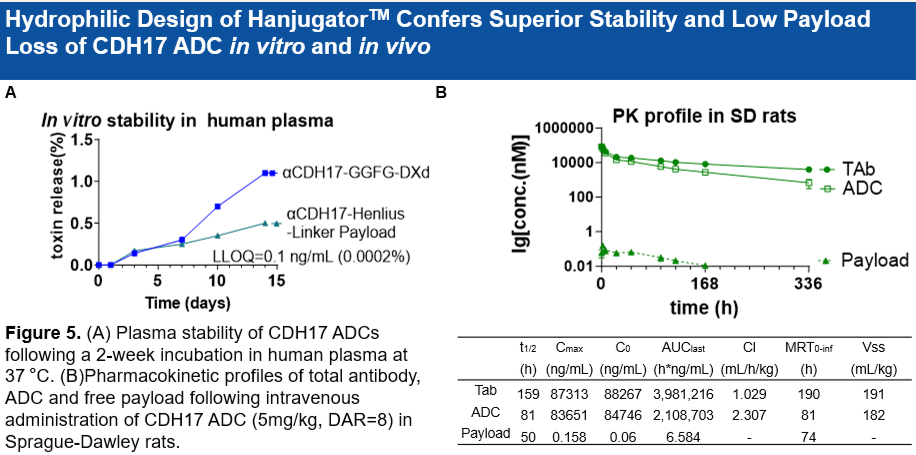
• 载荷(Payload)的活性与DXd相当,且具有强大的旁观者杀伤效应(Bystander Effect):该ADC的旁杀伤效应是基于deruxtecan ADC的19.7倍,有助于解决肿瘤的异质性问题。
3. 强效的体内抗肿瘤活性&耐药性肿瘤治疗潜力
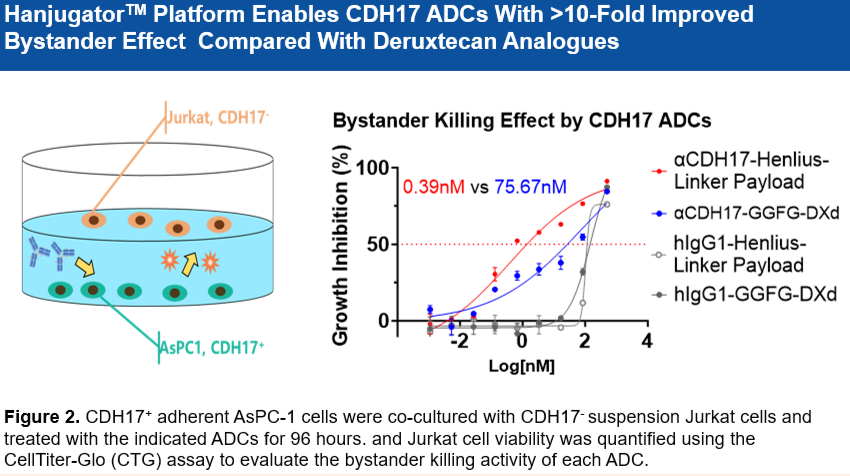
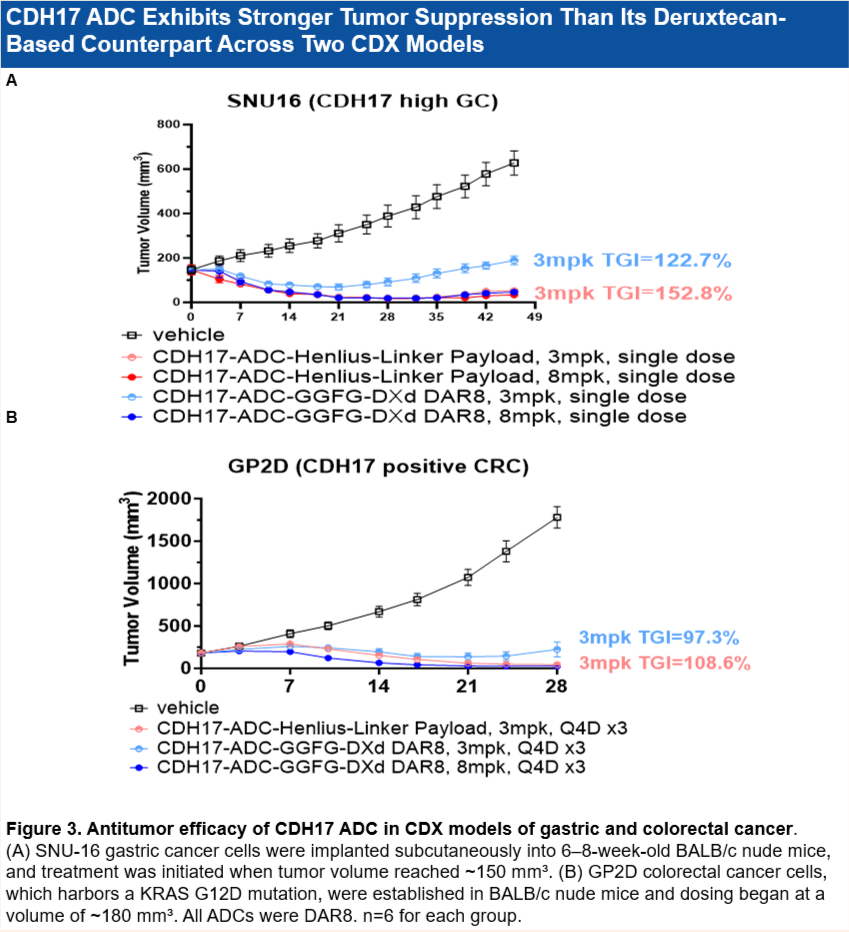
• 优于deruxtecan: 在多个CDX模型(如SNU16胃癌和GP2D结直肠癌)中,其疗效优于deruxtecan。
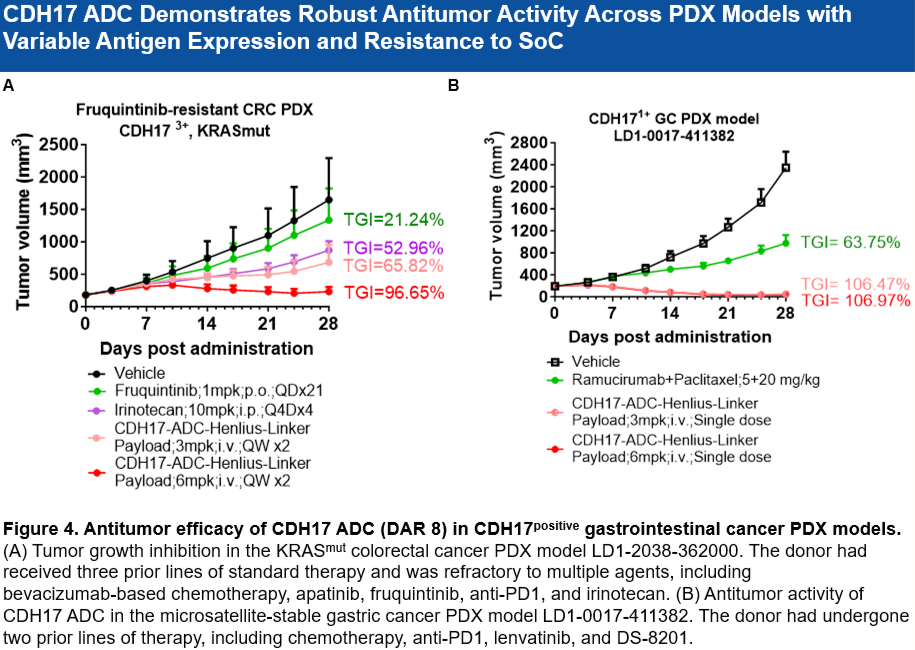
• 克服耐药性: 在对呋喹替尼(Fruquintinib)耐药的KRAS突变结直肠癌(CRC)PDX模型中显示出强劲疗效。
• 对低表达肿瘤有效: 在CDH17低表达、PD-L1低表达的胃癌(GC)PDX模型中,单次给药(3 mpk或 6 mpk)即可达到超100%的肿瘤生长抑制率(TGI)。
4. 潜在最优的安全性与耐受性
• 同类最优的安全性: 在非人灵长类动物(NHP)的毒理研究中,其最高非严重毒性剂量(HNSTD)达到40 mpk(Q3W x 3),在所有已知的同靶点ADC中处于最高水平。
• 良好的CMC特性: 抗体滴度高(>8 g/L),整体CMC属性良好。
综上所述,复宏汉霖的CDH17 ADC HLX403在临床前研究中展现了强效的抗肿瘤活性,同时在非灵长类动物中显示出同类最优的耐受性,有望突破肿瘤异质性和耐药性肿瘤的治疗瓶颈,为消化道肿瘤患者带来潜在更优的ADC治疗选择。该分子有望于2026年启动首次人体临床试验。
关于复宏汉霖
复宏汉霖(2696.HK)是一家国际化创新生物制药企业,致力于为全球患者提供高品质、可负担的生物药,产品覆盖肿瘤、自身免疫疾病、眼科疾病等领域。自2010年成立以来,公司已构建涵盖全球研发、临床、注册、生产及商业化的全产业链平台,拥有全球员工近4,000人,并在中国、美国和日本等多地设有运营及分支机构。依托生物类似药形成的稳健现金流反哺创新研发,复宏汉霖正稳步迈入“全球化2.0”阶段,持续打造可复制、可持续的全球增长模式。截至2026年初,公司共有10款产品在全球60个国家和地区获批上市,其中7款已在中国获批。在欧美主流生物药市场,复宏汉霖亦取得多项里程碑式突破,已有4款产品获得美国FDA批准、4款产品获得欧盟EMA上市授权,充分体现了公司在研发体系、质量管理及生产能力方面已全面对标国际最高标准。
在创新驱动方面,复宏汉霖依托上海、美国等多地协同布局的研发体系,构建了多元化、平台化的创新技术矩阵,覆盖免疫检查点抑制剂、免疫细胞衔接器(包括多特异性TCE)、抗体偶联药物(ADC)以及AI驱动的早期研发平台等前沿方向。目前,公司拥有50余项处于早期阶段的创新资产,其中约70%具备同类最佳(Best-in-Class)潜力,并在全球同步推进30余项临床研究。核心产品H药 汉斯状®(斯鲁利单抗,欧洲商品名:Hetronifly®)作为全球首个获批一线治疗小细胞肺癌的抗PD-1单抗,正加速全球布局,已在全球40余个市场获批上市;同时,多款潜力创新资产,包括PD-L1 ADC HLX43及新表位HER2单抗HLX22正全面推进全球关键性临床研究。依托通过中、欧、美三地GMP认证的生产体系,复宏汉霖已建成总产能达84,000升的生物药生产平台,形成覆盖全球六大洲的稳定供应网络。未来,复宏汉霖将始终坚持以患者为中心,聚焦未满足的临床需求,持续推动创新成果向临床价值与患者可及转化,在全球生物医药创新生态中创造长期而稳健的价值。
Henlius Debuts Preclinical Data of Its Proprietary CDH17 ADC, Showing a Differentiated Profile in GI Cancers at ADC Asia Congress 2026
The preclinical data presented at the conference highlight the molecule's key attributes:
-
Superior Antibody Performance and Specificity
-
High Affinity and Specificity: The antibody component of this ADC demonstrates nanomolar binding affinity to CDH17, with no off-target binding to other members of the cadherin family and exhibits a clean profile in MPA assays.
-
Differentiated Linker-Payload Platform (Hanjugator™)
-
Highly Hydrophilic Design: The proprietary hydrophilic linker-payload platform confers excellent druggability and developability.
-
Potent Anti-tumor Activity and Significantly Enhanced Bystander Effect: The payload exhibits anti-tumor activity comparable to DXd, while the ADC demonstrates a 19.7-fold stronger bystander killing effect than deruxtecan-based ADCs, a key feature for addressing tumor heterogeneity.
-
Robust In Vivo Anti-tumor Activity & Potential to Overcome Drug Resistance
-
Superior to Deruxtecan: Demonstrated superior efficacy compared to deruxtecan analogues across multiple cell line-derived xenograft (CDX) models, including SNU-16 gastric cancer cells and GP2D colorectal cancer cells.
-
Potential to Overcome Drug Resistance: Showed robust anti-tumor activity in a Fruquintinib-resistant, KRAS-mutant colorectal cancer patient-derived xenograft (PDX) model.
-
Efficacious in Low-Expressing Tumors: Achieved >100% tumor growth inhibition (TGI) with a single dose (3 mpk or 6 mpk) in a gastric cancer PDX model with low CDH17 and low PD-L1 expression.
-
Potential Best-in-Class Safety and Tolerability
-
Best-in-Class Tolerability Profile: In non-human primate (NHP) toxicology studies, the highest non-severely toxic dose (HNSTD) was established at 40 mg/kg (Q3W x 3), representing one of the highest levels reported for ADCs against the same target.
-
Favorable CMC Profile: Exhibits a high antibody titer (>8 g/L) and an overall favorable CMC profile.
In conclusion, Henlius' CDH17 ADC HLX403 has demonstrated potent anti-tumor activity in preclinical studies, along with a best-in-class tolerability profile in NHPs. It may help overcome therapeutic challenges, including tumor heterogeneity and drug resistance, offering a potentially improved ADC option for patients with gastrointestinal cancers. A first-in-human clinical trial for this molecule is expected to commence in the third quarter of 2026.
About Henlius
Shanghai Henlius Biotech, Inc. (2696.HK) is a global, innovation-driven biopharmaceutical company committed to delivering high-quality, affordable biologic therapies to patients worldwide. The Company focuses on major disease areas including oncology, autoimmune diseases, and ophthalmic diseases. Founded in 2010, Henlius has established an integrated, end-to-end biopharmaceutical platform encompassing global R&D, clinical operations, regulatory affairs, manufacturing, and commercialisation. The Company employs nearly 4,000 people globally and operates across multiple regions, including China, the United States, and Japan. Leveraging the stable cash flow generated from its biosimilar portfolio to support innovation, Henlius is steadily advancing into its “Globalisation 2.0” phase, building a scalable and sustainable global growth model. As of early 2026, Henlius has achieved regulatory approvals for 10 products across 60 countries and regions worldwide, including seven approvals in China. The Company has also reached multiple milestones in major biopharmaceutical markets, with four products approved by the U.S. Food and Drug Administration (FDA) and four products authorized by the European Medicines Agency (EMA), reflecting its globally aligned R&D capabilities, quality systems, and manufacturing standards.
Driven by innovation, Henlius has built a diversified, platform-based technology ecosystem through coordinated R&D efforts across Shanghai, the United States, and other regions. Its innovation platforms span immune checkpoint inhibitors, immune cell engager technologies (including multispecific T cell engagers), antibody-drug conjugates (ADCs), and AI-enabled early discovery platforms. The Company currently has more than 50 early-stage innovative assets, approximately 70% of which are expected to be best-in-class, with over 30 clinical trials ongoing globally. Henlius’ core product, serplulimab (trade name: Hetronifly® in Europe), is the world’s first anti–PD-1 mAb approved for first-line treatment of small cell lung cancer and has been approved in more than 40 markets worldwide with an accelerated globalisation process. In parallel, multiple high-potential innovative assets—including the PD-L1 ADC HLX43 and the novel epitope anti-HER2 mAb HLX22—are advancing through global pivotal clinical development. Supported by a biologics manufacturing network with a total capacity of 84,000L and GMP certifications from regulatory authorities in China, Europe, and the United States, Henlius has established a stable global supply system serving six continents. Guided by a patient-centred mission, Henlius remains focused on addressing unmet medical needs and translating scientific innovation into meaningful clinical value and patient access, contributing sustainably to the global biopharmaceutical ecosystem.
To learn more about Henlius, visit https://www.henlius.com/en/ and connect with us on LinkedIn at https://www.linkedin.com/company/henlius/.
References
-
Danpanichkul P, et al. Epidemiology of gastrointestinal cancers: a systematic analysis from the Global Burden of Disease Study 2021. Gut. 2024 Dec 10;74(1):26-34.
-
Singh A. Global burden of five major types of gastrointestinal cancer. Prz Gastroenterol. 2024;19(3):236-254.
-
Ntellas P, Chau I. Expanding the potential of antibody-drug conjugates in gastrointestinal malignancies: beyond HER2 targets. ESMO Gastrointest Oncol. 2025;8:100154.
-
Ge J, et al. A clinicopathological study on the expression of cadherin-17 and caudal-related homeobox transcription factor (CDX2) in human gastric carcinoma. Clin Oncol (R Coll Radiol). 2008 May;20(4):275-83.
-
Ito R, et al. Clinicopathological significant and prognostic influence of cadherin-17 expression in gastric cancer. Virchows Arch. 2005 Oct;447(4):717-22.










