关键研究启航,复星医药子公司复宏汉霖PD-L1 ADC HLX43在美国启动sqNSCLC II/III期临床试验

近日,复星医药子公司复宏汉霖(2696.HK)宣布,公司已在美国启动一项针对创新抗PD-L1 抗体偶联药物(ADC)HLX43用于晚期或转移性鳞状非小细胞肺癌(sqNSCLC)的 II/III期国际多中心临床研究(HLX43-NSCLC302)。该研究计划于完成II期部分后,与FDA沟通并启动后续的III期研究阶段,有望成为HLX43首个、同时也是其在肺癌领域的关键注册临床研究,为既往标准治疗失败后的sqNSCLC患者这一难治群体带来新的治疗选择。
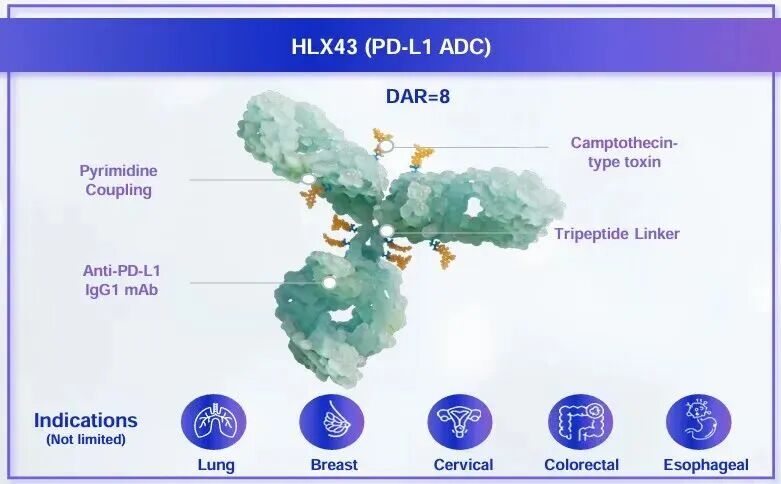
HLX43是一款潜在同类最优的广谱抗肿瘤PD-L1 ADC,兼具免疫检查点阻断与载荷细胞毒性的双重作用机制。目前,HLX43在NSCLC等实体瘤中展现出“高效、低毒”的初步临床疗效。根据2025 WCLC更新数据发布,在鳞状NSCLC的后线治疗中,HLX43已展现出初步的临床优势,接受2mg/kg剂量HLX43治疗的患者客观缓解率(ORR)达40.0%,疾病控制率(DCR)为73.3%。此外,针对多西他赛治疗失败的三线及后线NSCLC患者,HLX43的ORR仍达到30.0%,DCR为80%。HLX07是复宏汉霖自主开发的创新型抗EGFR的单抗,相比西妥昔单抗该产品具备更低的免疫原性和更好的靶点亲和力。根据2025 WCLC最新数据发布,HLX07联合斯鲁利单抗及化疗在EGFR高表达sqNSCLC患者一线治疗中展现出显著的抗肿瘤活性和持久疗效,在中位随访18.6个月时实现了约70%的客观缓解率(ORR)和超过90%的疾病控制率(DCR),且在高剂量组的中位无进展生存期(PFS)达到17.4个月。因此,HLX43有望协同抗EGFR单抗HLX07,发挥出较HLX43单药及抗EGFR单抗联合化疗更高的治疗潜力,进一步提升鳞状NSCLC等晚期实体瘤患者的治疗获益。
未来,复宏汉霖将持续聚焦患者未满足的临床需求,立足于HLX43等核心创新管线,不断放大产品的差异化治疗潜力,加速推动更大临床价值的释放,为全球患者带来更具突破疗效的治疗方案。
关于HLX43-NSCLC302
本研究为一项随机、开放标签、国际多中心 II/III 期临床研究,旨在评估HLX43(抗PD‑L1的ADC)单药或HLX43联合 HLX07(重组抗EGFR人源化单克隆抗体注射液),相较于多西他赛,在既往治疗失败的晚期/转移性鳞状非小细胞肺癌(NSCLC)患者中的 疗效和安全性。
该研究包括两个阶段。第一阶段为一项开放标签、随机、多中心的II期研究,符合入选标准的受试者将按照 1:1:1的比例随机分配至A组(HLX43 单药),B组(HLX43 联合 HLX07)或C组(多西他赛)。第二阶段为一项开放标签、随机、多中心的III期研究,将根据第一阶段结果选定 HLX43单药或HLX43联合HLX07作为试验组方案。计划入组约566例受试者,并按照1:1的比例随机分配至试验组(HLX43单药或HLX43联合HLX07)或对照组(多西他赛)。本研究研究主要目的是评估HLX43单药或HLX43联合HLX07对比多西他赛在经一线治疗失败的晚期鳞状NSCLC患者中的临床疗效,采用总生存期(OS)和无进展生存期(PFS)作为双主要终点。次要目的包括评价HLX43单药或HLX43联合HLX07的安全性、HLX43和HLX07的药代动力学(PK)特征、免疫原性,以及探索潜在的预测性或耐药性生物标志物。
关于复宏汉霖
复宏汉霖(2696.HK)是一家国际化创新生物制药企业,致力于为全球患者提供高品质、可负担的生物药,产品覆盖肿瘤、自身免疫疾病、眼科疾病等领域。自2010年成立以来,公司已构建涵盖全球研发、临床、注册、生产及商业化的全产业链平台,拥有全球员工近4,000人,并在中国、美国和日本等多地设有运营及分支机构。依托生物类似药形成的稳健现金流反哺创新研发,复宏汉霖正稳步迈入“全球化2.0”阶段,持续打造可复制、可持续的全球增长模式。截至2026年初,公司共有10款产品在全球60个国家和地区获批上市,其中7款已在中国获批。在欧美主流生物药市场,复宏汉霖亦取得多项里程碑式突破,已有4款产品获得美国FDA批准、4款产品获得欧盟EMA上市授权,充分体现了公司在研发体系、质量管理及生产能力方面已全面对标国际最高标准。
在创新驱动方面,复宏汉霖依托上海、美国等多地协同布局的研发体系,构建了多元化、平台化的创新技术矩阵,覆盖免疫检查点抑制剂、免疫细胞衔接器(包括多特异性TCE)、抗体偶联药物(ADC)以及AI驱动的早期研发平台等前沿方向。目前,公司拥有50余项处于早期阶段的创新资产,其中约70%具备同类最佳(Best-in-Class)潜力,并在全球同步推进30余项临床研究。核心产品H药 汉斯状®(斯鲁利单抗,欧洲商品名:Hetronifly®)作为全球首个获批一线治疗小细胞肺癌的抗PD-1单抗,正加速全球布局,已在全球40余个市场获批上市;同时,多款潜力创新资产,包括PD-L1 ADC HLX43及新表位HER2单抗HLX22正全面推进全球关键性临床研究。依托通过中、欧、美三地GMP认证的生产体系,复宏汉霖已建成总产能达84,000升的生物药生产平台,形成覆盖全球六大洲的稳定供应网络。未来,复宏汉霖将始终坚持以患者为中心,聚焦未满足的临床需求,持续推动创新成果向临床价值与患者可及转化,在全球生物医药创新生态中创造长期而稳健的价值。
Henlius Initiates a Phase 2/3 MRCT of Its Pan-Tumor PD-L1 ADC HLX43 for sqNSCLC in the US
Recently, Shanghai Henlius Biotech, Inc. (2696.HK) announced that the company has initiated a Phase 2/3 international multi-regional clinical trial (HLX43-NSCLC302) of HLX43, the company's innovative PD-L1-targeting antibody-drug conjugate (ADC) in patients with advanced or metastatic squamous non-small cell lung cancer (sqNSCLC) in the US. The study is planned to proceed to the subsequent Phase 3 stage following the completion of the Phase 2 stage and further discussions with the FDA. The Phase 3 stage is expected to serve as the first pivotal registration study for HLX43 and represents a significant milestone for HLX43 in the field of NSCLC, poised to offer a new therapeutic option for refractory sqNSCLC patients who have progressed on prior standard therapies.
Lung cancer is the most common cancer worldwide in terms of incidence and mortality. According to GLOBOCAN 2022, there were over 2.48 million new cases of lung cancer globally in 2022.1 Non-small cell lung cancer (NSCLC) is the predominant type of lung cancer (approximately 85%). By histology, NSCLC can be classified into squamous cell carcinoma (approximately 30%), adenocarcinoma (approximately 50%) and other subtypes, with a generally poorer prognosis for sqNSCLC.2 While immune checkpoint inhibitors (ICIs) combined with chemotherapy have significantly improved survival in advanced/metastatic sqNSCLC, effective options remain limited for patients progressing after first-line therapy. The current second-line and later-line treatment landscape is dominated by docetaxel-based regimens, with few agents demonstrating superior efficacy, highlighting a substantial unmet medical need.3-4 In recent years, antibody-drug conjugates (ADCs) have demonstrated favorable clinical efficacy in oncology, with promising activity observed in the later-line treatment of sqNSCLC.5-6 Separately, anti-EGFR monoclonal antibodies combined with chemotherapy have achieved progress in sqNSCLC, albeit primarily in EGFR-high expressing populations.7-9 The efficacy of novel therapeutic regimens incorporating anti-EGFR monoclonal antibodies in patients with low EGFR expression remains an area requiring further exploration.
HLX43 is a potentially best-in-class pan-tumor ADC candidate targeting PD-L1, which exhibits dual mechanisms integrating immune checkpoint blockade and payload-mediated cytotoxicity. Currently, it has demonstrated a manageable safety profile and encouraging efficacy in various solid tumors, with notable anti-tumor activity observed in multiple NSCLC patient subgroups. According to updated data presented at the 2025 WCLC, in the later-line treatment of sqNSCLC, HLX43 has demonstrated preliminary encouraging efficacy. Among patients receiving HLX43 at 2.0 mg/kg , the objective response rate (ORR) reached 40.0%, with a disease control rate (DCR) of 73.3%. Notably, in patients who had received prior docetaxel treatment, the ORR reached 30.0% with a DCR of 80.0%. HLX07 is an innovative anti-EGFR mAb developed by Henlius. Compared with cetuximab, HLX07 demonstrates lower immunogenicity and higher target affinity. According to updated results from the 2025 WCLC, the combination of HLX07 with serplulimab and chemotherapy demonstrated remarkable antitumor activity and durable efficacy in patients with EGFR-overexpressing sqNSCLC. At a median follow-up of 18.6 months, both dose groups achieved an objective response rate (ORR) of approximately 70% and a disease control rate (DCR) of over 90%. The median progression-free survival (PFS) reached 17.4 months in the high-dose group. HLX43 is expected to synergize with the anti-EGFR monoclonal antibody HLX07. This combination has the potential for superior efficacy compared to HLX43 monotherapy or an anti-EGFR antibody combined with chemotherapy, thereby offering greater clinical benefit to patients with advanced solid tumors such as sqNSCLC.
The company is currently accelerating the clinical development of HLX43. To date, over 700 patients have been enrolled globally in HLX43 studies, including more than 400 patients with NSCLC (over 60%). Among them, a multi-regional Phase 2 trial (HLX43-NSCLC201) is currently underway in China, the US, Australia, and Japan. Henlius has initiated over 10 clinical studies of HLX43, covering non-small cell lung cancer (NSCLC), cervical cancer (CC), esophageal squamous cell carcinoma (ESCC), head and neck squamous cell carcinoma (HNSCC), nasopharyngeal carcinoma (NPC), metastatic colorectal cancer (mCRC), gastric/gastroesophageal junction cancer (G/GEJ), pancreatic ductal adenocarcinoma (PDAC) and breast cancer (BC). Proof-of-concept (PoC) data in CC, ESCC, NPC, and HNSCC have been or will be presented at international conferences including ESMO Asia, ASCO GI, ASCO, and ESMO. Building on its intrinsic immuno-oncology (IO) activity, Henlius is exploring combination therapies, such as with the in-house anti-EGFR antibody HLX07 and anti-PD-1 mAb serplulimab, to maximize HLX43's clinical value.
Guided by its commitment to address unmet medical needs, Henlius will continue to advance its core pipeline including HLX43 to deepen the differentiated therapeutic potential of our products, accelerate the delivery of greater clinical value, and ultimately deliver more breakthrough treatment options to patients worldwide.
About HLX43-NSCLC302
This study is a randomized, open‑label, multi-center, global Phase 2/3 clinical study designed to evaluate the efficacy and safety of HLX43 (anti-PD‑L1 ADC) as monotherapy or in combination with HLX07 (a recombinant anti‑EGFR humanized monoclonal antibody injection) compared with docetaxel in patients with advanced or metastatic squamous non‑small cell lung cancer (NSCLC) who have failed prior therapy.
The study consists of two stages. Stage 1 is an open-label, randomized, multi-center phase 2 study, eligible subjects will be randomized in a 1:1:1 ratio to one of the following treatment arms: Group A (HLX43 monotherapy), Group B (HLX43 in combination with HLX07) or Group C (Docetaxel). Stage 2 is an open‑label, randomized, multi-center phase 3 study. Based on the results from Stage 1, either HLX43 monotherapy or HLX43 in combination with HLX07 will be selected as the investigational group. Approximately 566 subjects will be randomized in a 1:1 ratio to Investigational group (HLX43 monotherapy or HLX43 + HLX07) or Control group (Docetaxel).
The primary objective of this study is to evaluate the clinical efficacy of HLX43 monotherapy or HLX43 combined with HLX07 in the treatment of advanced squamous NSCLC after failure of first-line treatment, and the study adopts overall survival (OS) and progression‑free survival (PFS) as dual primary endpoints. Secondary objectives of this study include evaluation of safety of HLX43 monotherapy or HLX43 in combination with HLX07 vs. docetaxel, pharmacokinetic (PK) characteristics and immunogenicity of HLX43 and HLX07, and exploration of potential predictive or resistance biomarkers.
About Henlius
Shanghai Henlius Biotech, Inc. (2696.HK) is a global, innovation-driven biopharmaceutical company committed to delivering high-quality, affordable biologic therapies to patients worldwide. The Company focuses on major disease areas including oncology, autoimmune diseases, and ophthalmic diseases. Founded in 2010, Henlius has established an integrated, end-to-end biopharmaceutical platform encompassing global R&D, clinical operations, regulatory affairs, manufacturing, and commercialisation. The Company employs nearly 4,000 people globally and operates across multiple regions, including China, the United States, and Japan. Leveraging the stable cash flow generated from its biosimilar portfolio to support innovation, Henlius is steadily advancing into its “Globalisation 2.0” phase, building a scalable and sustainable global growth model. As of early 2026, Henlius has achieved regulatory approvals for 10 products across 60 countries and regions worldwide, including seven approvals in China. The Company has also reached multiple milestones in major biopharmaceutical markets, with four products approved by the U.S. Food and Drug Administration (FDA) and four products authorized by the European Medicines Agency (EMA), reflecting its globally aligned R&D capabilities, quality systems, and manufacturing standards.
Driven by innovation, Henlius has built a diversified, platform-based technology ecosystem through coordinated R&D efforts across Shanghai, the United States, and other regions. Its innovation platforms span immune checkpoint inhibitors, immune cell engager technologies (including multispecific T cell engagers), antibody-drug conjugates (ADCs), and AI-enabled early discovery platforms. The Company currently has more than 50 early-stage innovative assets, approximately 70% of which are expected to be best-in-class, with over 30 clinical trials ongoing globally. Henlius’ core product, serplulimab (trade name: Hetronifly® in Europe), is the world’s first anti–PD-1 mAb approved for first-line treatment of small cell lung cancer and has been approved in more than 40 markets worldwide with an accelerated globalisation process. In parallel, multiple high-potential innovative assets—including the PD-L1 ADC HLX43 and the novel epitope anti-HER2 mAb HLX22—are advancing through global pivotal clinical development. Supported by a biologics manufacturing network with a total capacity of 84,000L and GMP certifications from regulatory authorities in China, Europe, and the United States, Henlius has established a stable global supply system serving six continents. Guided by a patient-centred mission, Henlius remains focused on addressing unmet medical needs and translating scientific innovation into meaningful clinical value and patient access, contributing sustainably to the global biopharmaceutical ecosystem.
To learn more about Henlius, visit https://www.henlius.com/en/ and connect with us on LinkedIn at https://www.linkedin.com/company/henlius/.
References
-
Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-263.
-
Wang, Y., et al. Immunotherapy for advanced-stage squamous cell lung cancer: the state of the art and outstanding questions. Nat Rev Clin Oncol 22, 200–214 (2025).
-
中国临床肿瘤学会 中国临床肿瘤学会(CSCO). (2024)非小细胞肺癌诊疗指南
-
NCCN Guidelines Version 3. 2025, Non-Small Cell Lung Cancer.
-
Ahn MJ, et al. TROPION-Lung01 Trial Investigators. Datopotamab Deruxtecan Versus Docetaxel for Previously Treated Advanced or Metastatic Non-Small Cell Lung Cancer: The Randomized, Open-Label Phase III TROPION-Lung01 Study. J Clin Oncol. 2025 Jan 20;43(3):260-272.
-
Paz-Ares LG, et al. Sacituzumab Govitecan Versus Docetaxel for Previously Treated Advanced or Metastatic Non-Small Cell Lung Cancer: The Randomized, Open-Label Phase III EVOKE-01 Study. J Clin Oncol. 2024 Aug 20;42(24):2860-2872.
-
Pirker R, et al. EGFR expression as a predictor of survival for first-line chemotherapy plus cetuximab in patients with advanced non-small-cell lung cancer: analysis of data from the phase 3 FLEX study. Lancet Oncol. 2012 Jan;13(1):33-42.8.
-
Thatcher N, et al. Necitumumab plus gemcitabine and cisplatin versus gemcitabine and cisplatin alone as first-line therapy in patients with stage IV squamous non-small-cell lung cancer (SQUIRE): an open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2015 Jul;16(7):763-74.
-
Becotatug (JMT101, Beco) combined with docetaxel (albumin-bound, HB1801) for the treatment of patients (pts) with locally advanced squamous cell non-small cell lung cancer (sqNSCLC): A randomized control phase II study (BATTLE study). ESMO Asia 2025|2025-12-05.
联系方式

媒体:pr@fosunpharma.com
投资人:ir@fosunpharma.com










