全球研发 | 复星医药子公司复宏汉霖下一代TCE四特异性抗体HLX3901获国家药监局IND许可

2026年3月9日,复星医药子公司复宏汉霖(2696.HK)宣布,基于自主研发的创新型T细胞衔接器(T-cell engager, TCE)平台开发的创新分子——HLX3901(DLL3 x DLL3 x CD3 x CD28 四特异性抗体)的新药临床试验(IND)申请已获得国家药品监督管理局(NMPA)批准,拟用于晚期或转移性实体瘤的治疗,有望为临床难治性肿瘤,如小细胞肺癌(SCLC)等神经内分泌癌(NEC)带来突破性的免疫治疗方案。
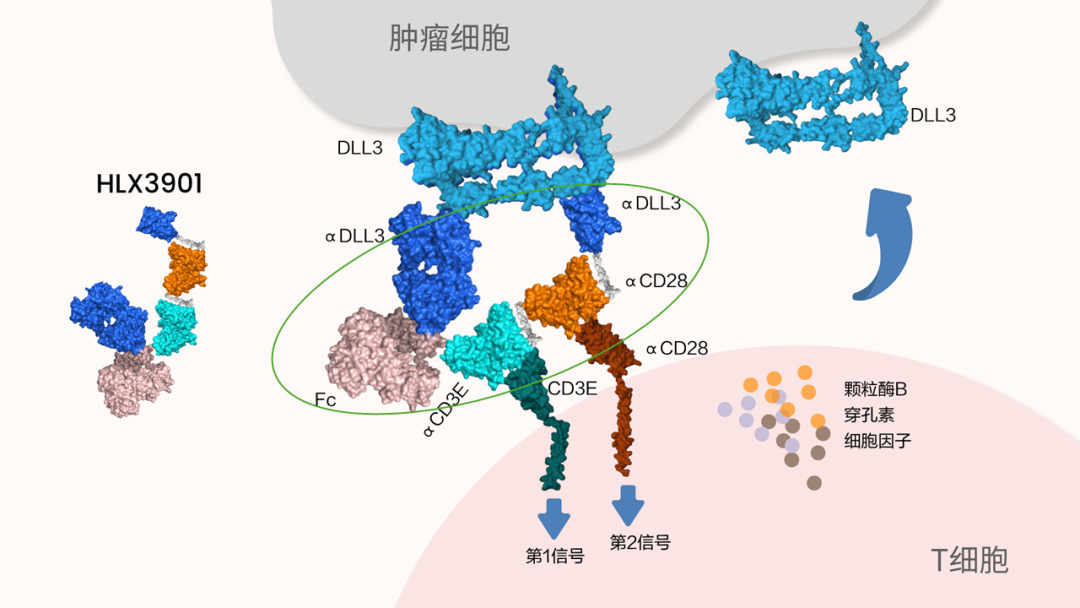
HLX3901是一款拥有自主知识产权的四特异性抗体药物,能够精准靶向DLL3双表位、CD3和CD28,作为新一代T细胞衔接器,HLX3901旨在增强对肿瘤细胞的杀伤,克服免疫抑制,并优化T细胞衔接器在实体瘤中的治疗窗口。其作用机制主要包括:
1)通过同时结合T细胞表面的CD3(第一信号) 和CD28 (共刺激信号),实现对T细胞的双重激活,从而增强T 细胞对DLL3阳性肿瘤细胞的靶向杀伤能力,提升抗肿瘤疗效;
2)通过协同激活T细胞第一信号与共刺激信号,促进T细胞的活化、增殖和存活能力,进而延长抗肿瘤免疫应答的持续时间,在低T细胞浸润的条件下依然展现出优异的抗肿瘤活性。
临床前研究显示,HLX3901在低效靶比条件下展现出更优的细胞毒性效应。在人类泛T细胞重构模型中,HLX3901比同类产品(如Tarlatamab)表现出更强且更持久的抗肿瘤活性。同时,在食蟹猴的初步毒性研究中,HLX3901耐受性良好,显示出较宽的治疗窗口。
作为复宏汉霖在免疫细胞衔接器领域的重要布局,HLX3901结合了公司AI驱动的智能药物设计与TCE平台技术,通过精巧的分子设计,该产品兼具持久的特异性T细胞激活、攻克低T细胞浸润肿瘤及显著降低细胞因子释放综合征(CRS)等多重优势,旨在克服第一代TCE在实体瘤治疗中的主要障碍。目前,公司已搭建起包括PD(L)1为核心的免疫检查点抑制剂平台、免疫细胞衔接器平台(如多特异性TCE平台)、Hanjugator™ADC 平台、AI驱动的一站式早期研发平台HAI Club在内的多维创新平台矩阵,在保障单个项目的研发质量与效率的同时,为打造具备全球竞争力的中长期创新管线提供了可持续的系统能力支撑。
深耕未满足的临床需求,公司加速扩充由高潜力分子组成的早期管线储备,多款产品于近期取得里程碑进展,HLX37(创新抗PD-L1/VEGF双特异性抗体)已获得IND批准用于晚期实体瘤治疗并高效完成首例患者给药;HLX701(新型SIRPα-Fc融合蛋白)凭借潜在更优的安全性,已启动在国内的II期临床研究,HLX97(新型KAT6A/B口服小分子抑制剂)也于近期同步获得CDE新药临床试验许可;更多临床前候选分子如HLX3902(STEAP1xCD3xCD28三特异性TCE)、HLX49(HER2双表位ADC)、HLX48(EGFRxcMET双抗ADC)、HLX105(一款抗体融合蛋白)在内的多个前沿分子也将加速迈向临床,为公司创新管线不断注入新鲜血液。
未来,复宏汉霖将继续坚持“以患者为中心”的研发理念,依托平台化、系统化的创新体系,加速推进具有全球竞争力的差异化创新管线布局,为全球患者提供更可及、更有效的治疗选择。
关于复宏汉霖
复宏汉霖(2696.HK)是一家国际化创新生物制药企业,致力于为全球患者提供高品质、可负担的生物药,产品覆盖肿瘤、自身免疫疾病、眼科疾病等领域。自2010年成立以来,公司已构建涵盖全球研发、临床、注册、生产及商业化的全产业链平台,拥有全球员工近4,000人,并在中国、美国和日本等多地设有运营及分支机构。依托生物类似药形成的稳健现金流反哺创新研发,复宏汉霖正稳步迈入“全球化2.0”阶段,持续打造可复制、可持续的全球增长模式。截至2026年初,公司共有10款产品在全球60个国家和地区获批上市,其中7款已在中国获批。在欧美主流生物药市场,复宏汉霖亦取得多项里程碑式突破,已有4款产品获得美国FDA批准、4款产品获得欧盟EMA上市授权,充分体现了公司在研发体系、质量管理及生产能力方面已全面对标国际最高标准。
在创新驱动方面,复宏汉霖依托上海、美国等多地协同布局的研发体系,构建了多元化、平台化的创新技术矩阵,覆盖免疫检查点抑制剂、免疫细胞衔接器(包括多特异性TCE)、抗体偶联药物(ADC)以及AI驱动的早期研发平台等前沿方向。目前,公司拥有50余项处于早期阶段的创新资产,其中约70%具备同类最佳(Best-in-Class)潜力,并在全球同步推进30余项临床研究。核心产品H药 汉斯状®(斯鲁利单抗,欧洲商品名:Hetronifly®)作为全球首个获批一线治疗小细胞肺癌的抗PD-1单抗,正加速全球布局,已在全球40余个市场获批上市;同时,多款潜力创新资产,包括PD-L1 ADC HLX43及新表位HER2单抗HLX22正全面推进全球关键性临床研究。依托通过中、欧、美三地GMP认证的生产体系,复宏汉霖已建成总产能达84,000升的生物药生产平台,形成覆盖全球六大洲的稳定供应网络。未来,复宏汉霖将始终坚持以患者为中心,聚焦未满足的临床需求,持续推动创新成果向临床价值与患者可及转化,在全球生物医药创新生态中创造长期而稳健的价值。
Henlius Announces IND Approval for Its Proprietary Next Generation Tetra-Specific T-Cell Engager HLX3901
Shanghai, China – March 9, 2026 – Shanghai Henlius Biotech, Inc. (2696.HK) today announced that the Investigational New Drug (IND) application for HLX3901, a novel tetra-specific antibody independently developed based on the company's proprietary T-cell engager (TCE) platform that targets DLL3xDLL3xCD3xCD28 , has been approved by the National Medical Products Administration (NMPA), for the treatment of advanced and metastatic solid tumors. It is expected to offer a breakthrough immunotherapy option for patients with neuroendocrine carcinoma(NEC)such as small cell lung cancer (SCLC).
DLL3 (Delta-like ligand 3), an inhibitory Notch ligand1-2, is highly expressed in approximately 80% of SCLC and neuroendocrine carcinoma3-5, while exhibiting minimal expression in normal tissues6-7. This profile establishes DLL3 as an ideal target for the treatment of SCLC and has driven the development of T-cell engagers (TCEs) as a research hotspot in targeted therapies. Currently, bispecific TCEs targeting DLL3 and CD3 have been approved 8 and have demonstrated promising therapeutic potential9. However, insufficient T-cell infiltration and the immunosuppressive state within the tumor microenvironment (TME) remain critical bottlenecks limiting the efficacy of TCEs10-11. Activation via CD3 alone (signal 1) in the absence of co-stimulatory signals (signal 2) can lead to T-cell anergy12, preventing durable and deep anti-tumor responses. CD28 is a key co-stimulatory molecule expressed on T cells. By binding to its ligands CD80 (B7-1) and CD86 (B7-2) on antigen-presenting cells (APCs), CD28 provides the essential second signal for full T-cell activation13-14.
HLX3901 is an innovative tetra-specific antibody with intellectual property, targeting dual epitopes of DLL3, along with CD3 and CD28. As a next-generation T-cell engager, HLX3901 is designed to enhance the cytolytic activity of T cells, overcome the immunosuppressive tumor microenvironment (TME), and optimize the therapeutic window of T-cell engagers in solid tumors. Its mechanism of action is primarily characterized by:
1)Dual activation of T cells through simultaneous engagement of CD3 (signal 1) and CD28 (co-stimulatory signal) on the T-cell surface, thereby enhancing the targeted killing of DLL3-positive tumor cells and improving anti-tumor efficacy.
2)Simultaneous engagement of CD3 and CD28 enhances T-cell activation, proliferation, and survival, promoting a prolonged T-cell response that drives potent anti-tumor activity even in tumors with low T-cell infiltration.
Preclinical studies indicate that HLX3901 exhibits enhanced cytotoxic effects at low effector-to-target ratios. In human pan-T cell reconstitution models, HLX3901 demonstrated stronger and more durable anti-tumor activity compared to reference molecules such as Tarlatamab. Furthermore, preliminary toxicity studies in cynomolgus monkeys showed that HLX3901 was well-tolerated with a broad therapeutic window.
HLX3901 represents a pioneering effort in Henlius' emerging immune cell engager pipeline. Developed through the synergistic integration of AI‑driven molecular design and the company’s T‑cell engager (TCE) platform, HLX3901 exemplifies a next‑generation TCE engineered to overcome key limitations of earlier constructs in solid tumors—achieving sustained and specific T‑cell activation, improved efficacy in the tumor microenvironment (TME) with low TIL density, and a reduced risk of cytokine release syndrome (CRS). The company has built a multidimensional innovation platform matrix, encompassing a PD(L)1-based checkpoint inhibitor platform, an immune cell engager platform (e.g.multi-specific TCEs), the proprietary Hanjugator™ ADC platform, and the AI-powered, all-in-one early-stage R&D platform HAI Club. This robust infrastructure ensures the quality and efficiency of individual projects while providing sustainable, systematic support for building a globally competitive mid-to-long-term innovative pipeline.
Rooted in addressing high-unmet medical needs, the company is efficiently expanding its early-stage pipeline with high-potential molecules, several of which have recently achieved significant milestones. HLX37 (an innovative anti-PD-L1/VEGF bispecific antibody) has received IND approval for advanced solid tumors and finished dosing its first patient. HLX701 (a novel SIRPα-Fc fusion protein) has initiated a Phase 2 clinical trial in China, leveraging its potentially improved safety profile. HLX97 (a novel oral small molecule KAT6A/B inhibitor) has recently received IND approval from the CDE. Furthermore, multiple preclinical candidates are accelerating towards the clinic, including HLX3902 (a STEAP1xCD3xCD28 trispecific antibody TCE), HLX49 (HER2xHER2 Novel Bi-paratopic ADC), HLX48 (an EGFRxcMET bispecific antibody ADC), and HLX105 (a fusion protein), continuously injecting new momentum into the company's innovation pipeline.
Looking ahead, Henlius will continue to adhere to its "patient-centric" R&D philosophy. Leveraging its integrated, platform‑based innovation engine, the company is committed to accelerating the development of a differentiated, globally competitive innovative pipeline, striving to provide more accessible and effective treatment options for patients worldwide.
About Henlius
Shanghai Henlius Biotech, Inc. (2696.HK) is a global, innovation-driven biopharmaceutical company committed to delivering high-quality, affordable biologic therapies to patients worldwide. The Company focuses on major disease areas including oncology, autoimmune diseases, and ophthalmic diseases. Founded in 2010, Henlius has established an integrated, end-to-end biopharmaceutical platform encompassing global R&D, clinical operations, regulatory affairs, manufacturing, and commercialisation. The Company employs nearly 4,000 people globally and operates across multiple regions, including China, the United States, and Japan. Leveraging the stable cash flow generated from its biosimilar portfolio to support innovation, Henlius is steadily advancing into its “Globalisation 2.0” phase, building a scalable and sustainable global growth model. As of early 2026, Henlius has achieved regulatory approvals for 10 products across 60 countries and regions worldwide, including seven approvals in China. The Company has also reached multiple milestones in major biopharmaceutical markets, with four products approved by the U.S. Food and Drug Administration (FDA) and four products authorized by the European Medicines Agency (EMA), reflecting its globally aligned R&D capabilities, quality systems, and manufacturing standards.
Driven by innovation, Henlius has built a diversified, platform-based technology ecosystem through coordinated R&D efforts across Shanghai, the United States, and other regions. Its innovation platforms span immune checkpoint inhibitors, immune cell engager technologies (including multispecific T cell engagers), antibody-drug conjugates (ADCs), and AI-enabled early discovery platforms. The Company currently has more than 50 early-stage innovative assets, approximately 70% of which are expected to be best-in-class, with over 30 clinical trials ongoing globally. Henlius’ core product, serplulimab (trade name: Hetronifly® in Europe), is the world’s first anti–PD-1 mAb approved for first-line treatment of small cell lung cancer and has been approved in more than 40 markets worldwide with an accelerated globalisation process. In parallel, multiple high-potential innovative assets—including the PD-L1 ADC HLX43 and the novel epitope anti-HER2 mAb HLX22—are advancing through global pivotal clinical development. Supported by a biologics manufacturing network with a total capacity of 84,000L and GMP certifications from regulatory authorities in China, Europe, and the United States, Henlius has established a stable global supply system serving six continents. Guided by a patient-centred mission, Henlius remains focused on addressing unmet medical needs and translating scientific innovation into meaningful clinical value and patient access, contributing sustainably to the global biopharmaceutical ecosystem.
To learn more about Henlius, visit https://www.henlius.com/en/ and connect with us on LinkedIn at https://www.linkedin.com/company/henlius/.
References
-
Kim JW, et al. DLL3 regulates Notch signaling in small cell lung cancer. iScience. 2022;25(12):105603.
-
Ladi E, et al. The divergent DSL ligand Dll3 does not activate Notch signaling but cell autonomously attenuates signaling induced by other DSL ligands. J Cell Biol. 2005;170(6):983-992.
-
Owen DH, et al. DLL3: an emerging target in small cell lung cancer. J Hematol Oncol. 2019;12(1):61.
-
Tanaka K, et al. Prevalence of Delta-like protein 3 expression in patients with small cell lung cancer. Lung Cancer. 2018;115:116–120.
-
Yao J, et al. DLL3 as an Emerging Target for the Treatment of Neuroendocrine Neoplasms. Oncologist. 2022 Nov 3;27(11):940-951.
-
Rudin CM, et al. Emerging therapies targeting the delta-like ligand 3 (DLL3) in small cell lung cancer. J Hematol Oncol. 2023;16(1):66.
-
Lobenhofer E, et al. P1.12-18 Nonclinical Safety Assessment of AMG 757, a DLL3 Bispecific T Cell Engager, in the Cynomolgus Monkey. Journal of Thoracic Oncology. 2019;14(10):S541.
-
Dhillon S. Tarlatamab: First Approval. Drugs. 2024;84(8):995-1003.
-
Ahn MJ, et al. Tarlatamab for Patients with Previously Treated Small-Cell Lung Cancer. N Engl J Med. 2023;389(22):2063-2075.
-
Hegde PS, Chen DS. Top 10 Challenges in Cancer Immunotherapy. Immunity.2020;52(1):17-35.
-
Belmontes B, et al. Immunotherapy combinations overcome resistance to bispecific T cell engager treatment in T cell-cold solid tumors. Sci Transl Med. 2021;13(608): eabd1524.
-
Schwartz RH. T cell anergy. Annu Rev Immunol. 2003;21:305-334.
-
Esensten JH, et al. CD28 Costimulation: From Mechanism to Therapy. Immunity. 2016;44(5):973-988.
-
Bhatia S, et al. Different cell surface oligomeric states of B7-1 and B7-2: implications for signaling. Proc Natl Acad Sci U S A. 2005;102(43):15569-15574.










